Molecular Machines: The Path to Simultaneous Diagnosis and Treatment
In his lab, he develops smart materials that encapsulate drugs, pioneering a groundbreaking approach that integrates diagnosis and treatment within a single molecule. Meet Dr. Abed Saady, an organic chemist who creates complex molecules in the lab that have never existed in nature, to harness them for the next revolution in medicine.
Imagine a world in which chemotherapy no longer comes with hair loss, nausea, or extreme weakness; a world in which the drug can navigate through the body like a guided missile and be released only when it reaches its precise target, without causing harm along the way. This vision, once considered almost impossible, is the scientific driving force behind Dr. Abed Saady, a senior lecturer in the Department of Chemistry and the Institute of Nanotechnology (BINA) at Bar-Ilan University, and one of the leading young researchers at the Dangoor Center for Personalized Medicine.
The Structure That Allows the Molecule to Move Freely Within the System
In 2016, four years before Saady began his postdoctoral work, the Nobel Prize in Chemistry was awarded for the field of designing and synthesizing molecular machines. These are small molecules engineered in the lab to perform controlled mechanical motion in response to an external stimulus such as light, heat, or a chemical change. Since then, researchers around the world have been exploring ways to apply these molecules across a range of fields.
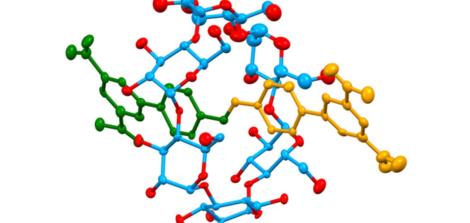
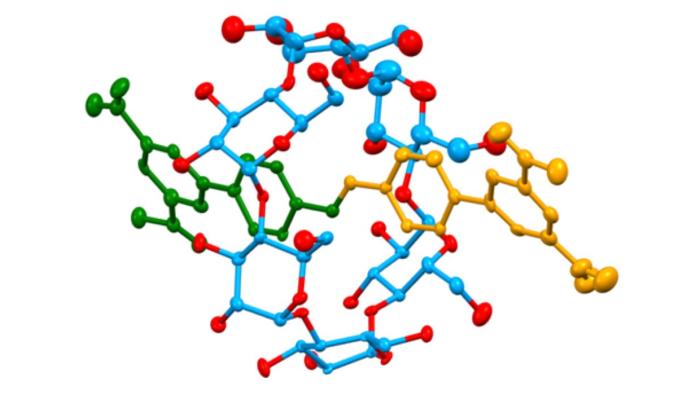
The central structure in which Dr. Saady specializes is called a rotaxane, a group of materials connected by a unique mechanical bond that enables them to perform chemical and biological functions like a machine. To understand how this machine works, imagine a tiny syringe with the drug inside, but with its plunger locked. For the drug to be released and take effect, someone must unlock it at precisely the right time and place.
Unlike a typical chemical bond, which acts like a strong glue holding atoms together in a rigid structure, a rotaxane is based on a mechanical bond. Imagine a ring threaded onto a rod, with stoppers at both ends preventing it from slipping off. “It’s like a key threaded onto a keyring,” Saady explains. “The key and the ring aren’t glued together; they’re simply physically interlocked, like links in a chain. That’s what allows the ring to slide freely along the rod from side to side without detaching.”
This mechanical freedom is what turns the molecule into a smart machine: the ring functions as a cap or a plunger. Dr. Saady engineers the molecule so that as it travels through the bloodstream, the ring sits in a position that blocks and conceals the drug beneath it. In this state, the drug remains “dormant” and does not harm healthy cells.
So, how is the lock opened? The secret lies in the use of triggers, stimuli unique to the disease environment. Dr. Saady designs the machine to respond to specific conditions found in a cancerous tumor, such as acidity levels or unique enzymes, while the lock remains tightly sealed throughout the rest of the body.
When the machine reaches its target, the ring “slides” in response to acidity or an enzyme, releasing the concealed drug. The molecular syringe releases its payload only at the target site, transforming the molecule from a static chemical structure into a dynamic machine that can read the body’s landscape and respond accordingly.
 “I“In the lab, the challenge is to produce only the ‘good’ form and not its mirror image. The ability to synthesize a machine with a specific chirality is critical to patient safety.”
“I“In the lab, the challenge is to produce only the ‘good’ form and not its mirror image. The ability to synthesize a machine with a specific chirality is critical to patient safety.”
Chemical Sensor: Seeing the Drug in Action in Real Time
The highlight of Dr. Saady’s research is theranostics, a field that combines diagnosis and treatment within a single molecule. This advanced approach uses the rotaxane as a multifunctional unit: it carries a fluorescent sensor or an MRI contrast agent alongside the therapeutic payload. “We engineer the material so that it ‘hides’ the drug within it and prevents it from interacting with receptors in the body on the way to the target,” Saady explains, describing how this innovation helps minimize side effects.
The rotaxane is equipped with a sensitive chemical sensor that detects conditions unique to a cancerous tumor, such as a specific acidity level (pH) or a particular enzyme. Only when the material reaches the tumor environment does the ring shift position or disassemble, releasing the drug with high precision. At the same time, the sensor “turns on” and signals to physicians: “I’ve reached the target and treatment has begun.” Instead of administering treatment blindly, doctors can observe how the drug is working in real time using MRI or fluorescence microscopy.
“As scientists, we need to be multitasking,” Saady says with a smile, “and the rotaxane is similar to us: it has different functions that can be modified simultaneously, yet it remains a single molecule.” For example, the ring can be engineered to break down in the presence of a high concentration of an enzyme specific to cancer cells, or under certain acidity conditions, such as those found in the stomach. “You can take one function and make it specific to a particular target,” he explains. “Any chemical modification you can think of can be tailored to our needs, and that includes personalized medicine in the future.”
 The laboratory of Dr. Saady at Bar-Ilan University. " I need my neighbors in biology and physics, and it’s a good thing we’re all under one roof.”
The laboratory of Dr. Saady at Bar-Ilan University. " I need my neighbors in biology and physics, and it’s a good thing we’re all under one roof.”
The Thalidomide Case: Molecular Direction That Makes All the Difference
As part of his postdoctoral work at the University of Southampton and the University of Birmingham in England, under the supervision of Dr. Steve Goldup, Dr. Saady gained extensive knowledge and experience in the synthesis of chiral rotaxanes. Chirality (from the Greek word for “hand”) refers to molecules that are mirror images of one another, much like our hands. Although they appear identical, they cannot be superimposed, and their effects on the body can be entirely opposite. “The body is a chiral environment; it can distinguish between a ‘right-handed’ and a ‘left-handed’ molecule,” he explains.
Because cells in the body respond differently to each molecular orientation, the ability to synthesize a machine with a specific chirality in the lab is critical for both treatment efficacy and patient safety.
To illustrate the importance of chirality, Saady points to one of the greatest drug-related disasters in history: the thalidomide case. “The drug was developed in the 1950s in Germany to treat pregnant women suffering from morning sickness,” he explains. “The problem was that it contained a mixture of two chiral molecules, mirror images of each other. While one form effectively treated nausea, its mirror image was toxic and led to a global tragedy: thousands of babies died at birth, and many others were born with severe deformities, without arms or legs.”
This lesson lies at the core of his work. “In the lab, the challenge is to produce only the ‘good’ form and not its mirror image. The ability to synthesize a machine with a specific directionality is critical to patient safety.”
Dr. Saady integrates in his work extensive experience in efficient synthesis, as reflected in a registered patent he holds from his doctoral research in the laboratory of Prof. Bilha Fischer, another leading researcher at the Dangoor Center. The development involves a synthetic method for producing a unique fluorescent material used to stain nucleoli in cells, a diagnostic tool that has already become commercial and serves many laboratories worldwide, enabling highly precise identification of pathological processes.
The Home of Science: Collaboration at the Dangoor Center
Dr. Saady repeatedly emphasizes that modern science is not the work of a single individual. “I’m a synthetic chemist, I specialize in making the material,” he says, “but for it to work, I need my neighbors in biology and physics, and it’s a good thing we’re all under one roof.” His work at the Institute of Nanotechnology (BINA) and the Dangoor Center enables daily interaction with researchers from different fields. “These collaborations, including with the nuclear medicine unit at Sheba Medical Center, are what turn research from theoretical to practical.”
Dr. Saady doesn’t ignore the challenges. “We’re not there yet,” he says candidly. “We have proof that we can build rotaxanes and break them down using specific enzymes, but now we’re working on optimizing the process.” The challenge isn’t only to create the molecular machine, but to do so with sufficient efficiency.
“In the past two years, we’ve managed to create three materials that had never been made before, using methods that were previously unknown. Those were very exciting moments, for me and for the students. But now we need to improve efficiency; instead of producing a single milligram, we need larger quantities that will allow us to move forward with the necessary testing.”
The use of advanced computer simulations (molecular dynamics) helps Dr. Saady “draw” these machines and test their stability even before reaching the test-tube stage. The computer runs simulations around the clock, checking with 95% accuracy whether the machine will break down along the way, saving valuable time for both the researcher and his students and allowing them to move forward more quickly.
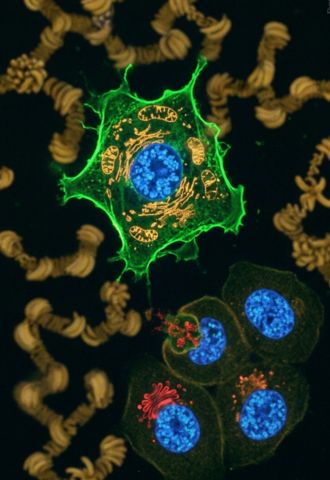 Production of a unique fluorescent material for staining nucleoli in the cell; a diagnostic tool that has become commercial, serves many laboratories worldwide, and ensures maximum accuracy in identifying pathological processes.
Production of a unique fluorescent material for staining nucleoli in the cell; a diagnostic tool that has become commercial, serves many laboratories worldwide, and ensures maximum accuracy in identifying pathological processes.
An Arab-Israeli Researcher During Wartime: An Island of Sanity
Dr. Saady represents the forefront of a new generation of young researchers who have grown at Bar-Ilan University, where he completed all his degrees with the highest honors. His return to the department as a senior faculty member in 2024 was especially moving. “It felt like coming home.”
Returning home also meant returning to family. Saady has two children, a son and a daughter, and his seven-year-old son joins him at the university when he has time off. Saady’s return to Israel from England in the midst of the war, in June 2024, was far from obvious. “Despite the natural concerns, as an Arab-Israeli researcher, I haven’t felt any change in how I’m treated at Bar-Ilan. Everyone welcomed me with open arms.” Saady, who decided from the outset to stay out of politics and focus on science, doesn’t let these things affect him. “I’m a man of science, that’s what interests me,” he says calmly. “I leave politics to the politicians.”
 Dr. Abed Saady: “We’ve achieved things that once seemed impossible. But nothing happens overnight...developing a drug takes 10 and 20 years.”
Dr. Abed Saady: “We’ve achieved things that once seemed impossible. But nothing happens overnight...developing a drug takes 10 and 20 years.”
A Vision for the Next Decade: Personalized Medicine for Every Patient
When asked about the future, Saady speaks about personalized medicine. “We’re working toward a future beyond one-size-fits-all treatments, where we can design a rotaxane tailored to each patient’s unique molecular profile, making therapy far more precise and effective. Nature already does this flawlessly; the ribosome is the perfect machine. We’re not trying to compete with nature, we’re learning from it.”
The path to a scientific breakthrough isn’t easy. “Sometimes you design a material that’s never existed before and you’re convinced it will work,” Saady says, “but then you try it again and again, and it just doesn’t.” Contrary to the romantic image of the lone scientist working in isolation, Saady emphasizes the importance of teamwork: “Together with my talented students, we’ve achieved things that once seemed impossible. But nothing happens overnight...developing a drug takes 10 to 20 years.”
He sees his role as a mentor as just as important as the research itself: “What matters most is raising a generation that truly cares about science, students who come to the lab each morning because they love what they do. In recent years, fewer students are choosing to pursue advanced degrees, and that’s our most valuable resource: curious, committed people. Even the best ideas won’t go anywhere without people to carry them forward.”
Since establishing his lab in October 2024, Saady has already achieved impressive results: eight students, including postdoctoral fellows, doctoral candidates, and master’s students, are working in the lab, and three scientific papers have already been published. “I’m very proud of that,” he says, “but we’re still in the development stage. Practical applications will come later.”
Saady, who arrives at the lab early in the morning and leaves late in the evening, sees his work as a mission: “My dream is to bring at least one real product to the pharmaceutical market, something that truly helps people. I’d like to know that one day, something I built made a difference. Until then, I’m focused on training the next generation of curious scientists, people who see chemistry as a way to build a better world.”
Last Updated Date : 27/04/2026