Genetic Matryoshka: Exploring the Viruses Hidden Within the Bacteria Inside Us

Billions of viruses inhabit our gut. Dr. Yishay Pinto, a principal investigator in the Faculty of Life Sciences at Bar-Ilan University and the Dangoor Center for Personalized Medicine, is developing computational tools that shed light on one of the greatest blind spots in modern medicine, the human microbiome, and exploring how this work could transform the way we diagnose and treat disease.
There’s a moment Dr. Yishay Pinto can still picture with absolute clarity. He’s sitting in a lecture hall, a young student, hearing for the first time about the human microbiome, about the trillions of microorganisms in our gut that shape nearly every aspect of our health. “I knew right then that this was what I wanted to study,” he says. “There was no question.”
Years later, that moment has come full circle. Back at Bar-Ilan University, where he trained as a scientist before heading to Stanford for his postdoctoral work, Dr. Pinto now leads his own lab. But while much of the field remains focused on gut bacteria, his work looks one layer deeper, at the viruses that live inside them, a hidden world that could redefine how we understand health and disease.
“We’re essentially a walking virus”
Dr. Pinto often describes the world he studies as a set of Russian nesting dolls. “There’s us, there are the bacteria that live inside us, and inside those bacteria, there are viruses.” The scale is hard to grasp. “We have more bacteria than human cells,” he says, “and far more bacterial genes than our own.” Then he adds, with a faint smile, “And there are even more viruses than bacteria. We’re essentially a walking virus.”
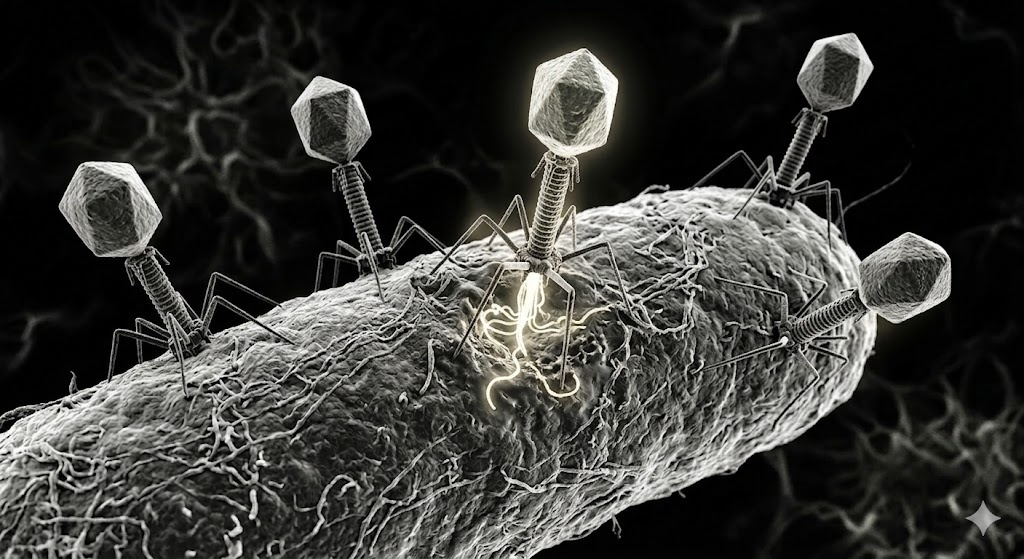
The viruses he studies are called bacteriophages, or simply phages. They don’t infect us directly, but the bacteria that live within us. “Just as there are viruses that infect humans, like herpes, HIV, or coronavirus, there are also viruses that infect bacteria,” he explains. “A phage is a piece of DNA wrapped in a protein shell. Under powerful microscopes, it looks and behaves like a tiny spacecraft. It lands on the surface of a bacterium, locks onto it with remarkable precision, and injects its genetic material inside. Sometimes it integrates into the host genome and lies dormant. Other times it takes over immediately, replicates itself, and kills the host.”
Taken together, these viruses form what’s known as the virome, the full collection of viruses in the human body. According to Dr. Pinto, it remains one of the last great blind spots in the life sciences.
Dark Matter: What Lives Within Us That We Still Don’t Understand
 “In our studies, we found a virus that appears in nearly every person we sampled – and it still doesn’t have a name.”
“In our studies, we found a virus that appears in nearly every person we sampled – and it still doesn’t have a name.”
The metaphor Dr. Pinto returns to is “dark matter.” In astronomy, most of the universe can’t be seen directly. He argues that something similar is happening inside us. Most of the viruses in our gut have never been grown in a lab, never isolated, and many don’t even have names.
“When we take a stool sample and sequence its DNA, we find that each of us carries hundreds of viruses that infect bacteria,” he says. “We have their genetic sequences, but beyond that, we know almost nothing about them. In one of my studies, we found a virus that appears in nearly every person we sampled, and it still doesn’t have a real name, just a random string of letters and numbers.”
The reason these viruses went largely unnoticed for so long is mostly technical. Isolating and growing even a single gut virus in the lab can take years, and there are hundreds of thousands of different viruses in the human gut alone. “It’s simply not feasible to study them using classical microbiology methods,” says Dr. Pinto. “That’s where researchers like me come in, people who know how to read DNA at scale.”
Phanta: A Tool for Detecting Viruses
To begin making sense of this hidden world, Dr. Pinto developed a tool called Phanta. He created it during his postdoctoral work at Stanford, and while the idea sounds straightforward, the computational leap is significant. Phanta takes sequencing data from a stool sample and identifies not only which bacteria are present, but also which viruses, and in what proportions.
“There were plenty of tools that could do this for bacteria,” he explains. “Phanta makes it possible to do the same for viruses, quickly and comprehensively, using the exact same sample.”
The challenge, as he describes it, is almost visual. “A stool sample is like a pile of Lego made up of pieces from a thousand different sets, and you have to put it all back together quickly, even though some of the pieces are missing.” His solution was to build a large database of known viruses and fine-tune the algorithm so it can recognize viral signatures alongside bacterial ones within the same dataset.
And the name? “It’s a nod to the drink,” he says with a smile. “There’s a bit of a habit in this field of naming algorithms after food and beverages.”
 “Based on bacterial profiles alone, we were able to predict quite accurately who would go on to develop gestational diabetes.”
“Based on bacterial profiles alone, we were able to predict quite accurately who would go on to develop gestational diabetes.”
The Gut Knows First: Predicting Gestational Diabetes Before It Happens
Back when his research was still focused on bacteria, Dr. Pinto, together with Prof. Omry Koren, a colleague at the Dangoor Center, set out to test an ambitious idea using stool samples from 400 pregnant women.
“Women are usually diagnosed with gestational diabetes toward the end of the second trimester, sometimes even at the start of the third,” he explains. “We took stool samples in the first trimester, about three months before the clinical diagnosis, and asked whether we could predict who would go on to develop the disease.”
The answer was yes. “We were able to predict, with fairly high accuracy, who would develop gestational diabetes, based on bacterial profiles alone.”
The implications go far beyond the study itself. Identifying risk early gives women a real window to act, adjusting diet and lifestyle in ways that can significantly reduce the risk for both mother and fetus. “The earlier you diagnose it, the more you can reduce the risks,” says Dr. Pinto. “What we showed is that even a simple stool sample can become a powerful predictive tool.”
This is where Dr. Pinto believes the next breakthrough will come. “Viruses are much more specific than bacteria,” he explains. “Their diversity is greater, which means they could potentially serve as more precise markers of disease. Our goal is to go back and do exactly what we did with gestational diabetes, but this time at the higher resolution the virome provides.”
That added layer may also help explain effects we’ve long observed but never fully understood. “When we take antibiotics, they change the population of bacteria in the gut, that’s well known,” says Dr. Pinto. “But there’s another effect we haven’t studied enough: dormant phages embedded in bacterial genomes suddenly wake up.”
The logic is simple. As antibiotics attack the host bacteria, the phages inside them sense that their host is under threat and switch into action. They replicate, infect other bacteria, and set off a chain reaction that reshapes the microbiome beyond the direct impact of the antibiotics themselves.
“We’re now studying how this activation of phages changes the population,” he says. “And it’s not just antibiotics. Even a change in diet, for example, can activate dormant phages. We’re still at the very beginning of understanding what this mechanism is doing to us.”
 “Vultures can eat carrion all day and nothing happens to them, while we’d get seriously ill or even die – what do they have that we don’t?”
“Vultures can eat carrion all day and nothing happens to them, while we’d get seriously ill or even die – what do they have that we don’t?”
From the Wild to the Lab: The Vultures That Taught Medicine
Before establishing his own lab, Dr. Pinto served as chief bioinformatician at Wild Biotech, an Israeli startup focused on the microbiomes of wild animals. The project was ambitious. “We worked with more than 200 species, sampling around 400 individual animals from remote locations across the globe, from Australia to the Falkland Islands, and reconstructed bacterial genomes that were completely unknown to science.”
“About three-quarters of the bacteria we identified from those samples were entirely new,” he says. One of the most striking discoveries came from vultures. “Vultures can eat carrion all day and nothing happens to them. If we did that, we’d get seriously ill or even die. So we asked, what do they have that we don’t?”
The answer lay in their microbiome. For example, a gene isolated from a bacterium in a vulture’s gut was found to break down botulinum toxin, the same toxin used, among other things, in Botox injections. “It shows that the microbiome of wild animals is like an open pharmacy we’ve only just begun to explore.”
AI Writing Genomes: The Lab’s Next Frontier
Dr. Pinto, 40, grew up in the Golan Heights and completed all his degrees at Bar-Ilan University. After a four-year postdoctoral fellowship at Stanford, he returned in the summer of 2023 to open his lab, at a particularly turbulent moment in Israel’s history. “It feels good,” he says simply. “I’m glad to have the chance to come back and do what I love.”
He now lives in Givat Shmuel with his wife and two children, just minutes from campus. The lab, opened less than a year ago, is already up and running, with two PhD students, four research students, a lab manager, and two additional researchers.
One of the most promising directions the lab is pursuing is the use of large language models, like ChatGPT, to study viral genomes. “A genome is a kind of language,” he explains. “It’s made up of four letters, and their order carries meaning. Just as a language model learns to read and write text, you can train a model to read and even generate genomic sequences.”
“The way we use language models in our research is a bit like taking a book, tearing out a page, and asking the computer to fill in the missing words based on context,” explains Dr. Pinto. “The model doesn’t just read the genome; it learns the rules that govern it, so it can infer what’s written on pages that are still hidden from us.”
In the lab, these models also help determine whether a virus tends to remain dormant, quietly embedded within a bacterial genome, or shifts into an active, lytic mode. But the implications go further. A lab in the United States recently demonstrated that a language model can be used to design a new phage, synthesize its DNA, insert it into a bacterium, and observe it functioning and infecting. “The idea is to use AI not just to read genomes, but to write them,” says Dr. Pinto. “And this isn’t science fiction anymore. It’s happening now.”
This points to the lab’s next central direction: not only diagnosing disease and identifying links between viruses and symptoms, but also designing tailored viral solutions based on a deep understanding. “Ultimately,” says Dr. Pinto, “we don’t just want to read this language, we want to start writing new chapters in it.”
.png) “We’re not claiming that phages will cure every disease – but the potential to predict, ease, and perhaps even prevent some of them is very real,” says Dr. Yishay Pinto.
“We’re not claiming that phages will cure every disease – but the potential to predict, ease, and perhaps even prevent some of them is very real,” says Dr. Yishay Pinto.
The Medical Vision: A Personalized Viral Cocktail
Where does all this lead? Dr. Pinto’s answer is ambitious. “I have a book in my office by Dr. Steffanie Strathdee, who saved her husband’s life using phage therapy,” he says. Phage therapy dates back to the early 20th century, before antibiotics, and is based on using phages to target specific pathogenic bacteria. When antibiotics emerged, the Western world largely abandoned the approach. In the past decade, however, with the rise of antibiotic resistance, interest has returned.
“Most phage research over the past few decades has focused on that question,” Dr. Pinto explains. “And that’s important, but it’s not what we do.” His lab is asking a different question: how to use phages to reshape the gut’s microbial ecosystem.
The idea builds on a practice that already exists: fecal transplants. It may sound unusual, but the process is simple. Stool is taken from a healthy donor with a rich, balanced microbial population, processed, and placed into capsules that the patient swallows like any other pill. The goal is to introduce a healthy bacterial community into the gut of someone whose microbiome has been disrupted or thrown out of balance.
Today, this treatment is used mainly in cases where harmful bacteria take over the gut and cause recurrent infections that antibiotics can no longer treat. A transplant from a healthy donor can restore microbial balance and prevent the infection from returning.
Dr. Pinto is now pushing the idea further. “We’re asking: what if we transplant only the viruses found in stool? They’re safer, and they may be just as effective.”
Looking ahead, he envisions labs capable of producing personalized phage cocktails for each patient, not only to target harmful bacteria, but also to fine-tune the microbiome for overall well-being, slow aging processes, treat inflammatory bowel diseases, and possibly even influence neurological conditions such as Parkinson’s and multiple sclerosis.
“The gut-brain axis is one of the most intensively studied areas today,” says Dr. Pinto. “Many of the molecules that affect the brain are produced by gut bacteria. We’ve seen increases in certain viruses in patients during flare-ups of multiple sclerosis. We’re not claiming that phages will cure every disease, but the potential to predict, ease, and perhaps even prevent some of them is very real.”
Last Updated Date : 27/04/2026